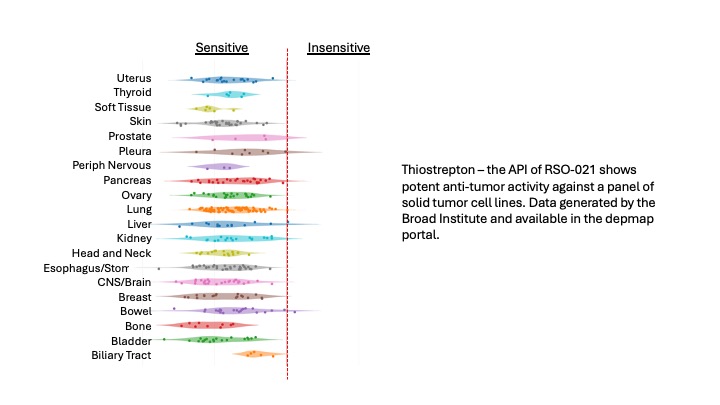
We are developing a portfolio of compounds that exploit disrupted metabolism and oxidative stress in aggressive cancers. Our pre-clinical discovery and testing programs utilize advanced cell, tissue and animal models to identify new candidate compounds to transition to IND enabling studies with the goal of bringing the most advanced and potent molecules to clinical studies.
Pipeline
Therapy
Indication
Discovery
IND Enabling
Phase 1
Phase 2
RSO-021
Intrapleural Monotherapy
Pleural Mesothelioma
NSCLC
NSCLC
RSO-021 + Paclitaxel
Intrapleural Combination
Metastatic Breast
Metastatic Ovarian
Metastatic Ovarian
RSO-023
Systemic
Solid Tumors
RSO-025
Topical
Melanoma
Other TBD
Other TBD